Chapter 1
Chemical Reactions And Equations
Some Important Headings and Its Defination with Example
1. Chemical Reaction
A chemical reaction is the process by which reactants react with each other to yield products. In chemical reactions, the reactant molecules are completely different from the molecules of the products formed.
Example
Hydrogen gas reacts with oxygen gas to form Water as product

2. Chemical Equation
A chemical Equation is a written Representation of a Chemical Reaction
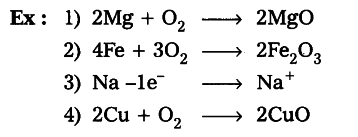
Types of Chemical Reactions
There 4 types of chemical Reactions which are given below
(1) Combination Reaction
A combination reaction is a reaction where two or more elements or compounds combine to form a single compound. Such reactions are represented by equations of the following form:
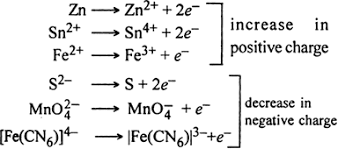
Example
Magnesium reacts with Oxygen to form Magnesium Oxide
2Mg + O2 ————–> 2MgO
Combination Reaction is Also Known As Synthesis Reaction.
Some more example of Combination Reaction
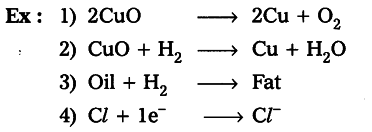

2. Decomposition Reaction
Decomposition is a type of chemical reaction. It is defined as the reaction in which a single compound splits into two or more simple substances under suitable conditions
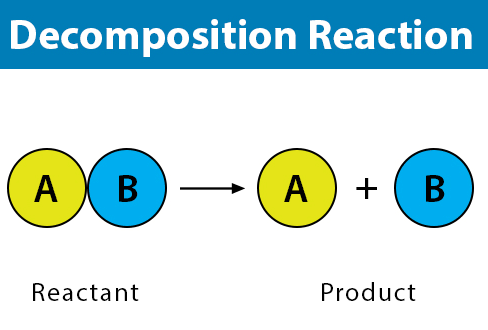
Example
Breaking of Sodium Chloride into Sodium and Chlorine
Nacl ————–> Na+Cl
Some more Example of Decompostion Reaction
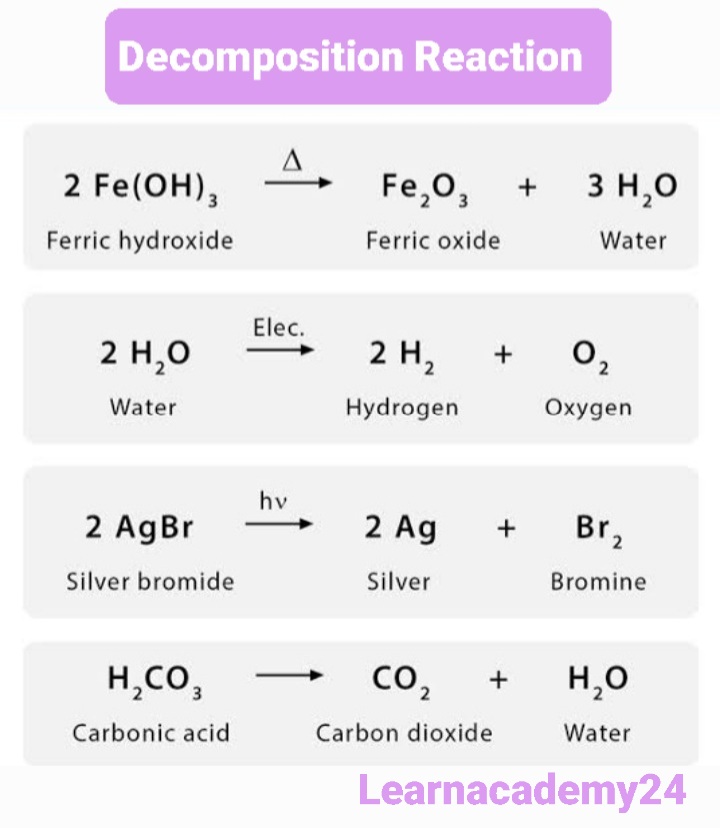
Note:- The Decomposition Reaction May be Either Exothermic or Endothermic.for Example
- The decomposition of NO to N2 & O2 is exothermic.
- Decomposition of ozone (O3) to oxygen (O2) is exothermic.
3. Displacement Reaction
Single displacement reactions are reactions where one reactant replaces part of the other:
AB + C → AC + B
An example is the reaction between iron and copper sulfate to produce iron sulfate and copper:
Fe + CuSO4 → FeSO4 + Cu
Some more Example of Displacement Reaction

4. Double Displacement Reaction
Those reactions in which two compounds react by an exchange of ions to form two new compounds are called double displacement reactions. In double replacement reactions, the positive ions exchange negative ion partners. Many double displacement reactions occur between ionic compounds that are dissolved in water. A double replacement reaction is represented by the general equation
We can write as
Example
AB+CD ———-> AD+BC
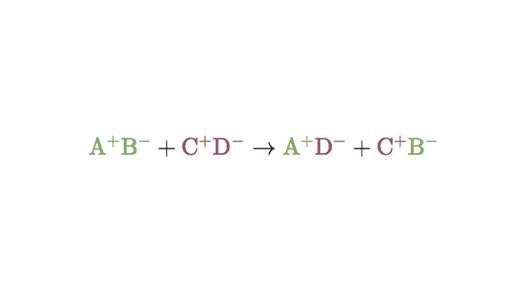
Example
BaCl2 +2AgNO3 ——— 2Agcl +Ba( NO3 ) 2
Some more Example of Double Displacement Reaction

* Oxidation
The Addition of oxygen to a substance or removal of Hydrogen from a substance know as oxidation.
Example
2Mg + O2 ——— 2MgO
* Reduction
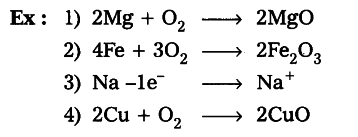
The removal of Oxygen from a substance or Addition of hydrogen to a substance known as Reduction
Example
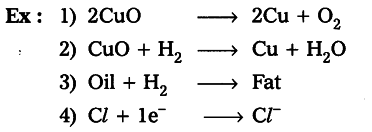
Reducing Agent
A substance which loses electrons to other substances in a redox reaction and gets oxidized to the higher valency state is called a reducing agent
Example

Oxidising Agent
An oxidizing agent is a substance in a redox chemical reaction that gains or “accepts”/”receives” an electron from a reducing agent. In other words, an oxidizer is any substance that oxidizes another substance
Example
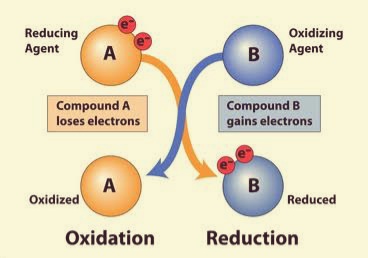
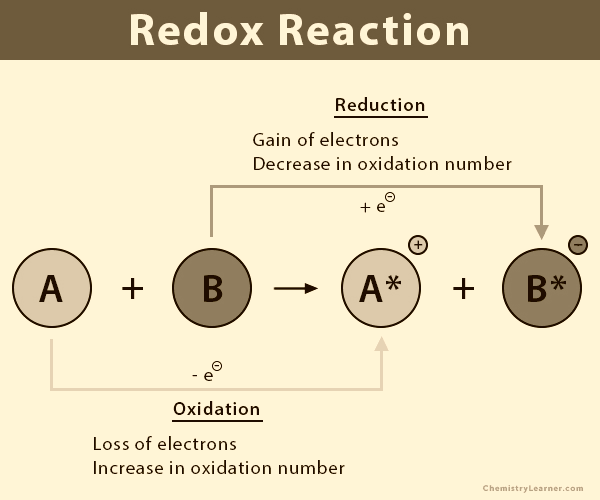
Redox Reaction
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. An oxidation-reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron. Redox reactions are common and vital to some of the basic functions of life, including photosynthesis, respiration, combustion, and corrosion or rusting
Example